Woman Sues Birth Control Company After Discovering ‘Lime-Sized’ Brain Tumor at 30
A Long Island mom says her life changed in an instant when doctors found a brain tumor “the size of a lime.” Now, she’s taking her fight to court.
Elizabeth Fleurisma has filed a lawsuit against Pfizer, alleging an injectable birth control she used for years did not adequately warn about a possible increased risk of certain tumors.
The case is one of many lawsuits that have been filed in recent months, with plaintiffs making similar claims about the same product.
A diagnosis that reshaped her daily life
Fleurisma says she was 30 when she learned she had a large brain tumor. She later underwent an extensive surgery that lasted around 16 hours, followed by weeks of radiation therapy.
Doctors removed much of the tumor, but Fleurisma says a portion remains lodged in her skull.
In interviews reported by US outlets, she described ongoing challenges after treatment, including struggling with speech and feeling disoriented in familiar environments.
What the lawsuit claims
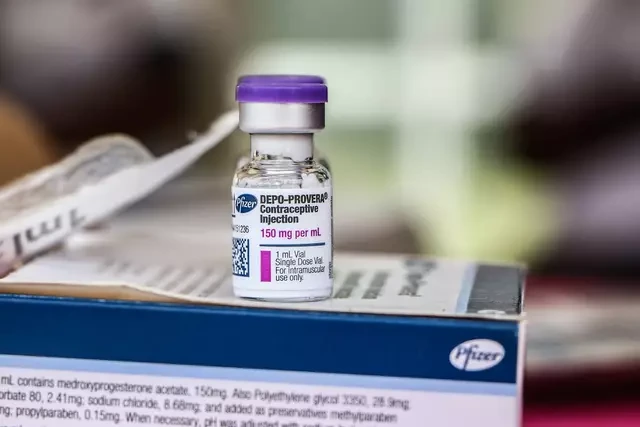
The lawsuit targets Pfizer, the manufacturer of Depo-Provera, an injectable contraceptive typically administered every three months.
Fleurisma alleges the product’s labeling and warnings did not clearly communicate what she believes is a heightened risk of developing intracranial meningiomas. These tumors often grow slowly and form in the meninges, the membranes that cover the brain and spinal cord.
The filings also claim Pfizer knew—or should have known—about the alleged risk and failed to provide stronger warnings earlier. As with any civil complaint, these claims represent the plaintiff’s allegations and have not been proven in court.
Why she chose an injection over the pill
Fleurisma’s account describes a common decision many patients face: convenience and consistency.
After giving birth as a teenager, she reportedly looked for a birth control option that did not require remembering a daily pill. Depo-Provera, given by injection, became her choice.
According to the report, she used the injections for about eight years.
How Depo-Provera works
Depo-Provera contains progestin, a synthetic form of a hormone involved in the reproductive cycle.
Progestin-based contraceptives can prevent pregnancy by stopping ovulation and making it harder for sperm to reach an egg. Injectable birth control appeals to some people because it offers longer coverage between doses than daily tablets.
The lawsuit’s focus is not on whether the drug prevents pregnancy, but on whether the product label sufficiently warned users about a specific safety concern.
Pfizer’s response and what comes next
Pfizer has pushed back against the accusations, saying it believes the claims lack merit and that it will defend itself in court.
In statements reported by the media, the company has said it stands behind the safety and efficacy of Depo-Provera and notes it has been used by millions of women worldwide.
The wider legal fight includes far more than Fleurisma’s case. Reports have described hundreds—potentially more—lawsuits connected to the same alleged issue, with plaintiffs seeking damages and stronger accountability around risk communication.
What this story signals for patients and regulators
Cases like this often reignite a broader conversation about how drug risks get communicated to the public, especially when a product has remained on the market for decades.
In situations involving widely used medicines, health agencies like the FDA play a major role in evaluating safety data and updating labeling when new evidence emerges. Media reports have also referenced a Reuters account that Pfizer sought additional warning language after reviewing research in 2023 that suggested a potential link.
For now, the core issue will play out in court: what the available evidence shows, what Pfizer knew and when, and whether warnings met legal and regulatory standards.
Featured Image Credit: Getty Stock Image